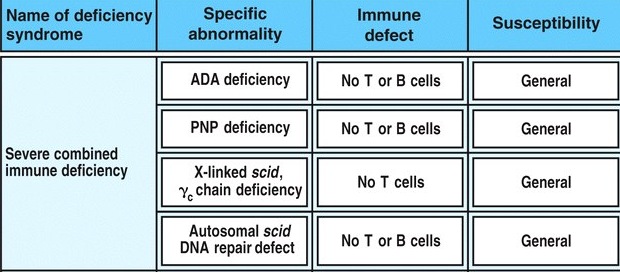
Long-term outcomes after gene therapy for adenosine deaminase severe combined immune deficiency - ScienceDirect
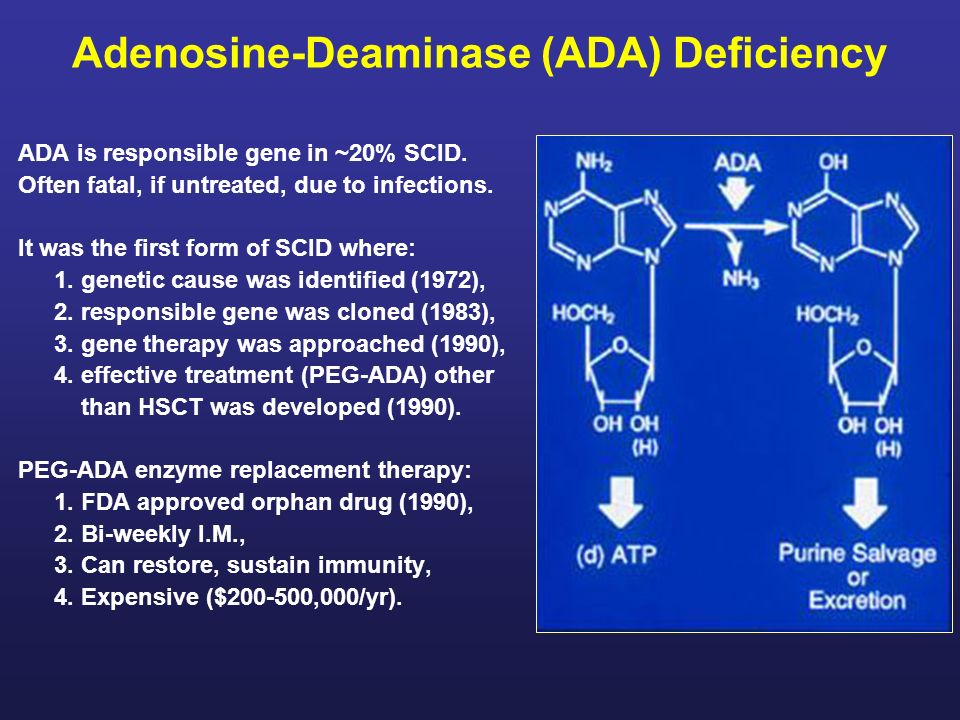
Adenosine-Deaminase (ADA) Deficiency ADA is responsible gene in ~20% SCID. Often fatal, if untreated, due to infections. It was the first form of SCID. - ppt download

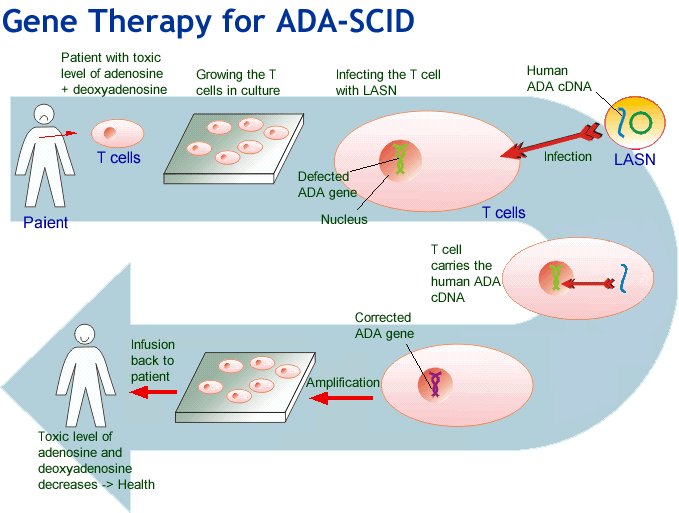
Gene delivery using AAV8 in vivo for disease stabilization in a bimodal gene therapy approach for the treatment of ADA-deficient SCID - ScienceDirect

Illustration Treatment Severe Combined Immunodeficiencyscid Using: Stock-Vektorgrafik (Lizenzfrei) 1959146710 | Shutterstock